The beginning of almond harvest is upon us in the Sacramento Valley, and while the bulk of the in-season pest management decisions are behind us, now is the time for a key activity in your year-round IPM Program – harvest samples.
Obtaining harvest samples and taking the time to crack out and identify the sources of damage is well worth the investment. Grade sheets alone will not distinguish among the various types of insect damage. Knowing the culprit provides valuable information, such as: How successful was my pest management program this season? How effective were my treatment decisions, timings, materials, and applications? What might I need to do differently next year?
Information from annual harvest samples should be kept to establish block-specific historical records, which help focus pest management activities in subsequent years. With worm damage, noting the size and stage of development (larvae, pupae, emerged pupae) indicates when the initial infestation occurred and can be compared with monitoring and treatment records to improve future pest management decisions.
The main insect pests responsible for much of the damage observed at harvest include: navel orangeworm (NOW), peach twig borer (PTB), oriental fruit moth (OFM), ants, and plants bugs (leaffooted bug and stink bugs). If worms are present in your samples, they can be visually distinguished by the following key characteristics:
NOW: Larvae milky white to pinkish with brown head capsule. Look for the dark crescent shapes on second segment behind the head capsule on either side.

Navel orangeworm larva and pupa.

Crescent-shaped marking that is diagnostic of navel orangeworm larvae.
PTB: Larvae brown and white striped with dark head capsule.

Peach twig borer larva and pupa.
OFM: Larger larvae pinkish with brown head capsule (newly hatched larvae are whitish with black head capsule). Somewhat similar in appearance to NOW but without crescent-shaped markings. A hand lens can be used to detect presence of an anal comb on the last abdominal segment.

Oriental fruit moth larva.
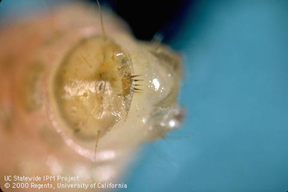
The anal comb on the last abdominal segment is diagnostic of oriental fruit moth.
Ideally, a sample of 500 nuts per block is the UC IPM guideline for harvest samples. However, you know your orchards best. Obtain a representative sample of nuts from each block at harvest. Collect nuts from multiple areas of the block. If you are collecting after nuts have been blown into rows, make sure to collect a stratified sample (not just nuts sitting on the top layer of the rows – infested nuts may weigh less and be a larger proportion of those on the top layer). Larger blocks may require more samples to provide an accurate estimate of damage identification. However, smaller blocks with a great deal of variation in pest pressure may require more samples than larger, more uniform blocks. Multiple years of sampling and site-specific historical records can help guide your harvest sampling process.
In a perfect world, samples should be processed soon after collecting. However, storing harvest samples and cracking out once things slow down is often necessary. At a minimum, prior to storing for any length of time, refrigerate the samples, remove hulls within a few days of collection, and record hull infestation. This will limit rotting and movement of pests between nuts. Once hulls are off, relatively dry nuts will hold for a period of time in cold storage (refrigerator temperatures); however, freezer temperatures are best for longer term storage or when seeking to identify pest developmental stage.
The table below summarizes some key distinctions for determining the likely source of insect damage in harvest samples. Larger photos of the insect damage are shown below the table.
| KERNEL DAMAGE | FRASS | WEBBING | BORING | FEEDING BEHAVIOR | ||
| NAVEL ORANGEWORM | Deep chewing into nut |  | Often quite a lot | Yes | Yes | Feed in groups |
| PEACH TWIG BORER | Shallow channels & surface groove on kernels |  | No | No | No | Generally solitary |
| ORIENTAL FRUIT MOTH | Shallow channels and surface groove on kernels |  | Very little, reddish-brown | No | No | Feed in groups |
| ANTS | Scraping or peeling of kernel skin, deep hollowing of nut, "sawdust" present |  | No (don't confuse "sawdust" for frass) | No | Hollowing | Varies |
| PLANT BUGS | Dark spots on the kernel |  | No | No | No | Piercing-sucking |

NOW damage.

PTB damage.

OFM damage.

Ant damage.

Leaffooted bug damage.
Note: Originally posted August, 2017. Updated August, 2018.


Leave a Reply